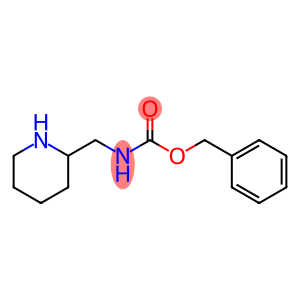
benzyl piperidin-2-ylmethylcarbamate hydrochloride
PIPERIDIN-2-YLMETHYL-CARBAMIC ACID BENZYL ESTER
CAS: 184044-09-5
Molecular Formula: C14H20N2O2
benzyl piperidin-2-ylmethylcarbamate hydrochloride - Names and Identifiers
| Name | PIPERIDIN-2-YLMETHYL-CARBAMIC ACID BENZYL ESTER |
| Synonyms | 2-CBZ-aMinoMethyl piperidine-HCl benzyl piperidin-2-ylmethylcarbamate Benzyl piperidine-2-ylmethylcarbamate benzyl N-(piperidin-2-ylMethyl)carbaMate Piperidine-2-ylmethylcarbamic acid benzyl ester PIPERIDIN-2-YLMETHYL-CARBAMIC ACID BENZYL ESTER benzyl piperidin-2-ylmethylcarbamate hydrochloride PIPERIDIN-2-YLMETHYL-CARBAMIC ACID BENZYL ESTER-HC,L |
| CAS | 184044-09-5 |
| InChI | InChI=1/C14H20N2O2/c1-16(13-9-5-6-10-15-13)14(17)18-11-12-7-3-2-4-8-12/h2-4,7-8,13,15H,5-6,9-11H2,1H3 |
benzyl piperidin-2-ylmethylcarbamate hydrochloride - Physico-chemical Properties
| Molecular Formula | C14H20N2O2 |
| Molar Mass | 248.32 |
| Density | 1.081±0.06 g/cm3(Predicted) |
| Boling Point | 409.2±28.0 °C(Predicted) |
| pKa | 12?+-.0.46(Predicted) |
| Refractive Index | 1.557 |
benzyl piperidin-2-ylmethylcarbamate hydrochloride - Introduction
PIPERIDIN-2-YLMETHYL-CARBAMIC ACID benzoyl ESTER, also known as piperidinyl benzoate, is an organic compound. Its chemical formula is C12H15N3O2 and its molecular weight is 233.27g/mol.
Nature:
PIPERIDIN-2-YLMETHYL-CARBAMIC ACID benzoyl ESTER is a white crystalline solid with a faint peculiar odor. It is soluble in water, ethanol and Ester solvents at room temperature. Its melting point is about 145-150 degrees Celsius. The common crystalline form of this compound is a hydrate, comprising a monohydrate of one water molecule and a dihydrate of two water molecules.
Use:
PIPERIDIN-2-YLMETHYL-CARBAMIC ACID BENZYL ESTER is a widely used drug intermediate and reagent. It is widely used in the synthesis of a variety of pharmaceutical products, such as anti-influenza drugs, antihypertensive drugs, antipsychotic drugs and sedatives. In addition, it can be used as an ingredient in pesticides and cosmetics.
Method:
PIPERIDIN-2-YLMETHYL-CARBAMIC ACID benzoyl ESTER can be prepared by various methods, including propionamide method and acetamide method. Among them, the propionamide method is to dissolve piperidine in acetone, and then react with chlorinated formamide to generate the target product. In the acetamide method, piperidine is dissolved in ethanol and then reacted with chloroacetate to form the product.
Safety Information:
PIPERIDIN-2-YLMETHYL-CARBAMIC ACID benzoyl ESTER is irritating and may cause irritation to the skin, eyes and respiratory tract. During use and processing, it is necessary to avoid direct contact, if necessary, wear protective glasses, gloves and masks. In addition, during storage and transportation, care should be taken to avoid contact with oxidants, strong acids and strong bases to prevent dangerous reactions. When using this compound, follow local safe practices. If contact with skin or eyes occurs, rinse immediately with plenty of water and seek medical attention.
Nature:
PIPERIDIN-2-YLMETHYL-CARBAMIC ACID benzoyl ESTER is a white crystalline solid with a faint peculiar odor. It is soluble in water, ethanol and Ester solvents at room temperature. Its melting point is about 145-150 degrees Celsius. The common crystalline form of this compound is a hydrate, comprising a monohydrate of one water molecule and a dihydrate of two water molecules.
Use:
PIPERIDIN-2-YLMETHYL-CARBAMIC ACID BENZYL ESTER is a widely used drug intermediate and reagent. It is widely used in the synthesis of a variety of pharmaceutical products, such as anti-influenza drugs, antihypertensive drugs, antipsychotic drugs and sedatives. In addition, it can be used as an ingredient in pesticides and cosmetics.
Method:
PIPERIDIN-2-YLMETHYL-CARBAMIC ACID benzoyl ESTER can be prepared by various methods, including propionamide method and acetamide method. Among them, the propionamide method is to dissolve piperidine in acetone, and then react with chlorinated formamide to generate the target product. In the acetamide method, piperidine is dissolved in ethanol and then reacted with chloroacetate to form the product.
Safety Information:
PIPERIDIN-2-YLMETHYL-CARBAMIC ACID benzoyl ESTER is irritating and may cause irritation to the skin, eyes and respiratory tract. During use and processing, it is necessary to avoid direct contact, if necessary, wear protective glasses, gloves and masks. In addition, during storage and transportation, care should be taken to avoid contact with oxidants, strong acids and strong bases to prevent dangerous reactions. When using this compound, follow local safe practices. If contact with skin or eyes occurs, rinse immediately with plenty of water and seek medical attention.
Last Update:2024-04-09 20:52:54
Supplier List
Product Name: PIPERIDIN-2-YLMETHYL-CARBAMIC ACID BENZYL ESTER Request for quotation
CAS: 184044-09-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 184044-09-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: PIPERIDIN-2-YLMETHYL-CARBAMIC ACID BENZYL ESTER Request for quotation
CAS: 184044-09-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 184044-09-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
View History
